It occurs as carbonates magnesite, MgCO 3, and dolomite, CaMg (CO 3. Its cosmic abundance is estimated as 9.1 × 10 5 atoms (on a scale where the abundance of silicon 10 6 atoms). The physical and chemical properties of the metals and nonmetals may be used to tell them apart. Magnesium is the eighth most abundant element in Earth ’s crust (about 2.5 percent) and is, after aluminum and iron, the third most plentiful structural metal. 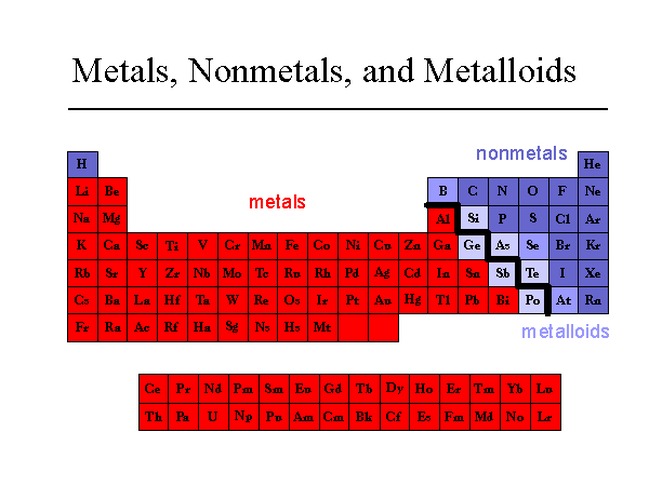
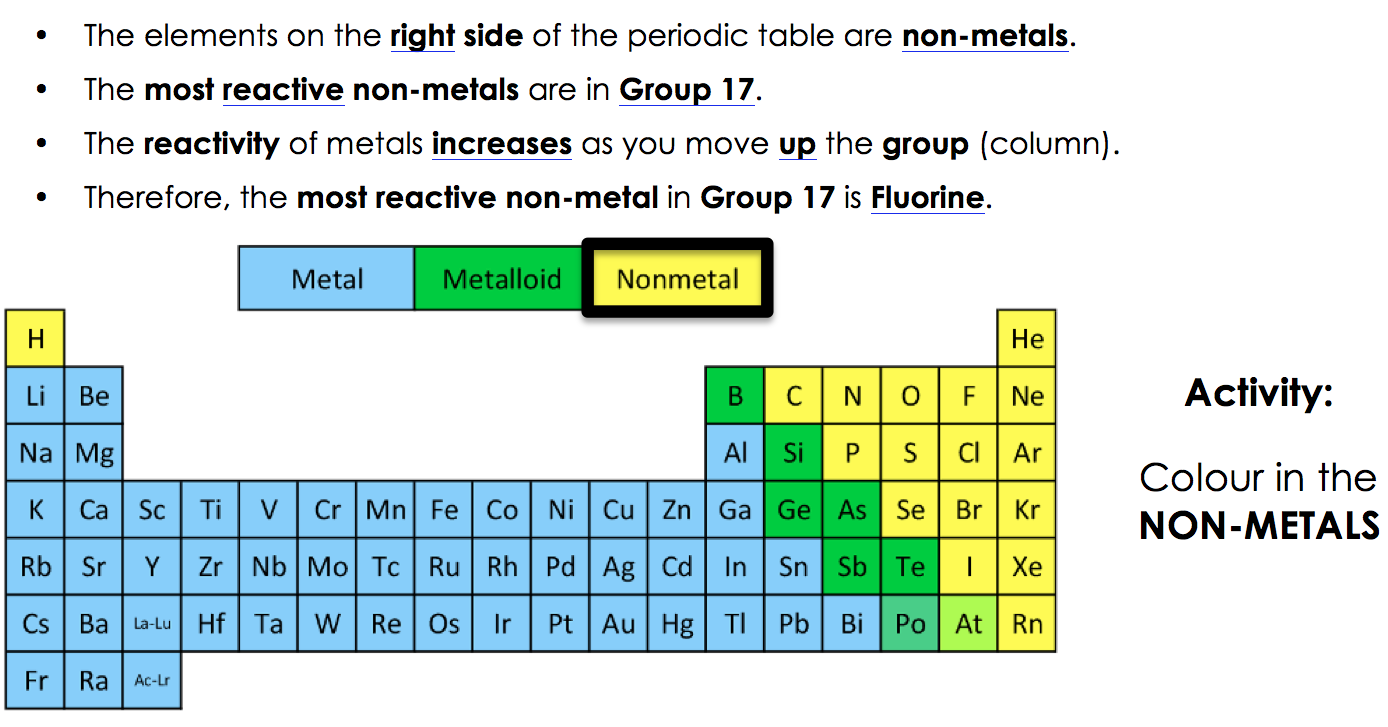
Elements just to the left of the line may be termed metalloids or semimetals and have properties intermediate between those of the metals and nonmetals. These elements and those to the right of them are nonmetals. On the periodic table, metals are separated from nonmetals by a zig-zag line stepping through carbon, phosphorus, selenium, iodine, and radon. This includes the alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. They have lower melting and boiling points than most metals and aren't usually good conductors. Table 3.1 Element Symbol Type ofHardness Malleability Ductility Conducts Sonority surface Electricity On the bases of the observations recorded in Table 3. View Available Hint (s) Reset Help Metal Nonmetal Semimetal KB. 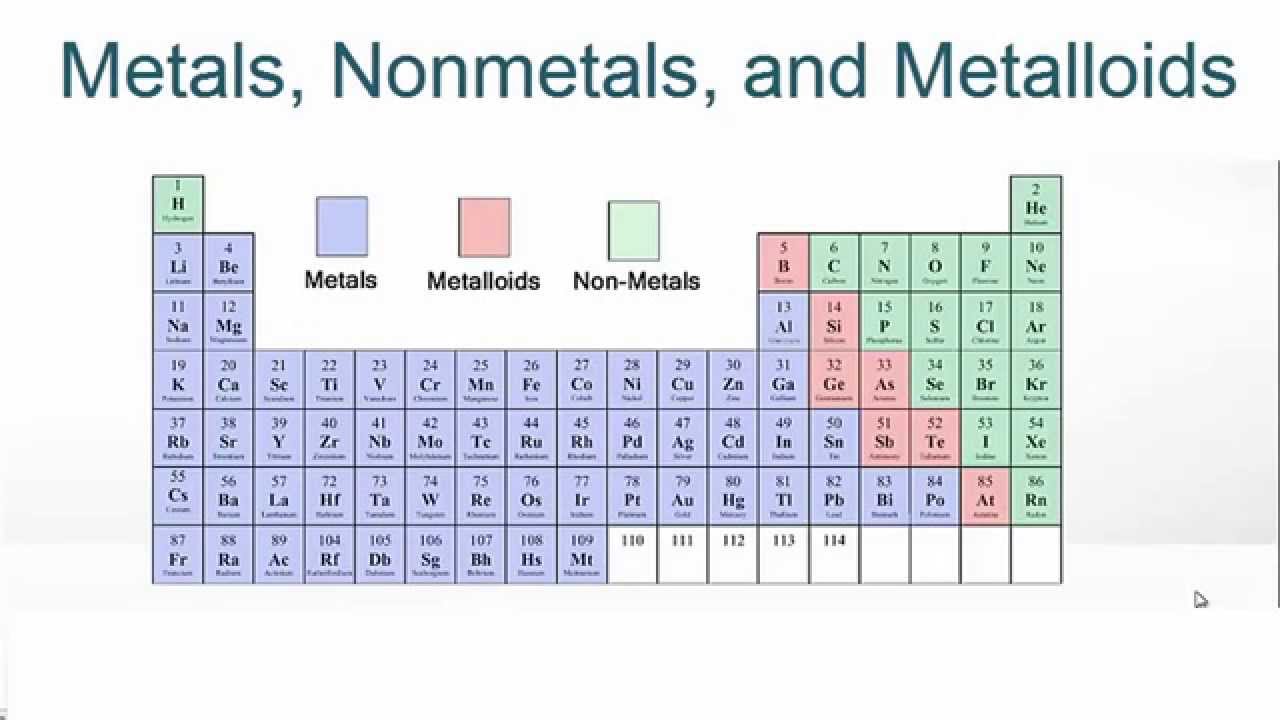
Drag the appropriate items to their respective bins. Question: Classify each element as a metal, nonmetal, or semimetal. Compile your observations regarding metals and non-metals in Table 3.1. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Nonmetals tend to be softer, often colorful elements. n Carry out the Activities 3.1 to 3.4 and 3.6 with these non-metals and record your observations.Magnesium lies to the left of the diagonal line marking the boundary between metals and nonmetals, so it should be a metal. Metals tend to be hard, metallic-looking solids, with high electrical and thermal conductivity values and high melting and boiling points. Elements can be divided into metals and non-metals, and it is important to know whether a particular element is a metal or a non-metal. In Figure 5.1.1, selenium lies above and to the right of the diagonal line marking the boundary between metals and nonmetals, so it should be a nonmetal.Element Symbol Atomic Number Color Luster Metal or Nonmetal aluminum carbon copper iron nitrogen magnesium manganese nickel oxygen phosphorus potassium silicon gold silver tin zinc cobalt lead chlorine Osmium cesium antimony 26 C. The periodic table consists of elements that are metals, those that are nonmetals, and elements with properties intermediate between the two groups (metalloids). 25 B PHYSICAL PROPERTIES OF THE ELEMENTS Observe the elements in the Periodic Table display and fill in the following table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
